Reducing blood pressure to target levels is a major priority in preventing clinical events in hypertension. Although there are many possible combinations of BP-lowering therapies, the best combination for the black population is still a subject of debate. Furthermore, current guideline recommendations are inconsistent and conflicting for black patients. CREOLE stands for (Comparison of Three Combination Therapies in Lowering Blood Pressure in Black Africans). The study aims to produce evidence that would help in the development of clinical guidelines for the treatment of hypertension in sub-Saharan Africa.
The CREOLE trial will be performed at ten investigational sites in six countries in Sub-Saharan Africa (green colour), including Cameroon. CREOLE compares the efficacy of three different combinations of antihypertensive therapies (amlodipine/hydrochlorothiazide, amlodipine/perindopril, and perindopril/hydrochlorothiazide) in black Africans. It is a randomised, single-blind, multicentre, parallel group, three-arm superiority trial. Participants will be randomised to one of the three treatments.
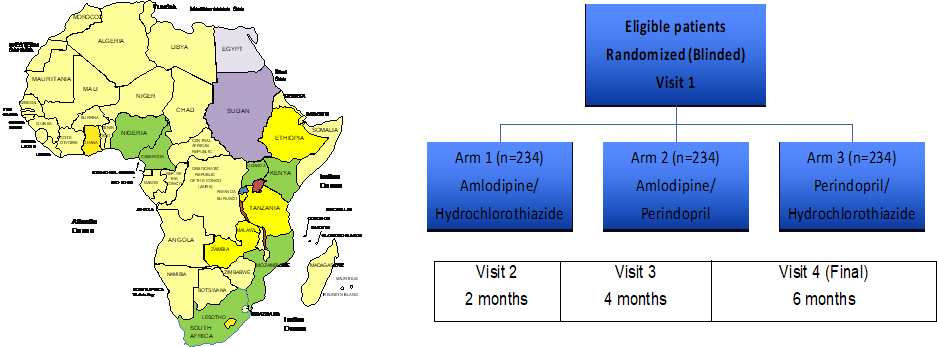
The primary outcome measure is change in ASBP from baseline to 6 months. This will be calculated as the difference between the mean ASBP at randomization and that at the end of follow up.
Secondary endpoints include a change in the following from baseline to 6 months:
- 24 hour ADBP
- Clinic systolic and diastolic BP
- Daytime and night time BP
- BP variability measured by ASBP
- Proportion of patients who achieve BP control (defined as clinic BP < 140/90 mmHg) at two, four and six months
- Proportion of “responders” (defined as clinic BP reduction > 20mmHg SBP and >10 mmHg DBP) at two, four and six months.
- Micro- and macro-albuminuria
- Fasting blood glucose
- Fasting blood lipid profile
The Principal Investigator is Dr Dike Ojji, University of Abuja, Nigeria. The CREOLE trial is implemented and executed in Cameroon by the CRENC team under the leadership of Dr Anastase Dzudie. The CT identifier number is NCT02742467 .
Sponsor: University of Abuja.
More info at https://clinicaltrials.gov/ct2/show/NCT02742467
Dr. Dike Ojji, (Principal Investigator) explains CREOLE Clinical Trial in an interview with AIM HY.